Lattice energy equation for nacl1/20/2024   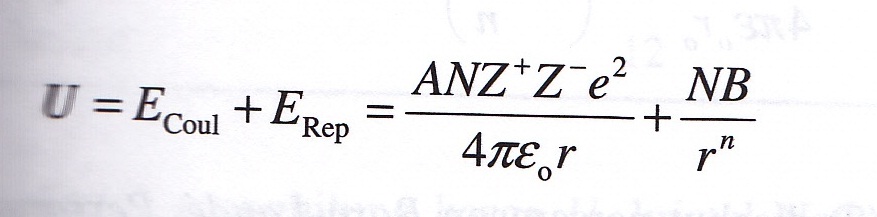
The internuclear distance in the gas phase is 175 pm.Īnswer: −3180 kJ/mol = −3. Rearranging the equation to solve for lattice energy yields the following: Lattice energy Heat of formation- Heat of atomization- Dissociation. The lattice energy of the sodium chloride solid U H f 0. Consequently, in accordance with Equation 4.1.1, much more energy is released when 1 mol of gaseous Li +F − ion pairs is formed (−891 kJ/mol) than when 1 mol of gaseous Na +Cl − ion pairs is formed (−589 kJ/mol).Ĭalculate the amount of energy released when 1 mol of gaseous MgO ion pairs is formed from the separated ions. Here in the Born Haber cycle for NaCl, except for the lattice energy, all the other enthalpies can be measured experimentally. The Born exponent is typically between 5 and 12.\( \newcommand/mol\right )=-891\ kJ/mol \) īecause Li + and F − are smaller than Na + and Cl − (see Figure 3.2.7 ), the internuclear distance in LiF is shorter than in NaCl. For example, CF is 439 kJ/mol, CCl is 330 kJ/mol, and CBr is 275 kJ/mol. Lattice energy of nacl Lattice Energy - Division of Chemical Education, Purdue University WebLattice energy, U, is defined as the enthalpy required to. The Born–Landé equation gives an idea to the lattice energy of a system. When one atom bonds to various atoms in a group, the bond strength typically decreases as we move down the group. Lattice Energy Lattice Energy Formula Example 1: Compute the Lattice energy of NaCl by using Born-Lande equation. 
This expression takes into account both the Born interactions as well as the Coulomb attractions. Born forces are one type of force that acts upon atoms in an ionic lattice. de 2023 Here, we can observe that MgOs lattice energy is significantly higher than NaCl. E = − N A M z + z − e 2 4 π ε 0 r 0 ( 1 − 1 n ) Calculated lattice energies The Born-Landé equation is a concept originally formulated in 1918 by the scientists Born and Landé and is used to calculate the lattice energy (measure of the strength of bonds) of a compound. 6.13C: Born Forces is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by LibreTexts. In 1918 Max Born and Alfred Landé proposed that the lattice energy could be derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. The equation derived by Bom gives for the lattice energy of a NaCl crystal the value -762kJ/mol (i.e. U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an. The salient features of its structure are: Chloride ions are ccp type of arrangement. Explain born haber cycle to calculate lattice energy of nacl enthalpies of solution and hydration - chemguide Veeb8. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contain 39.34 g Na and 60.66 g Cl. 
The Born–Landé equation is a means of calculating the lattice energy of a crystalline ionic compound. The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Equation 4.1: U kQ1Q2 r0, where U > 0 (4.2.1) (4.2.1) U k Q 1 Q 2 r 0, w h e r e U > 0. Sodium chloride, also known as salt or halite, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
